NEW RESEARCH SUGGESTS STATE BANS ON ENDS PRODUCTS IN THE UNITED STATES MAY HAVE UNINTENDED CONSEQUENCES, INCLUDING INCREASES IN CIGARETTE SALES
Research & Analysis
September 21, 2020
Unacceptable levels of underage use have eroded public trust in our industry, leading to the introduction of well-intentioned state-level product bans that may have the unintended consequence of turning adult smokers away from alternatives and back to combustible cigarettes.
As a part of the Premarket Tobacco Product Application process, Juul Labs has built up its science and evidence-based capabilities with a comprehensive research program focused on examining the public health impact of JUUL System. Our population impact team, in particular, explores the impact of policies and interventions in the United States and globally in order to understand how they can help reduce underage use of nicotine products.
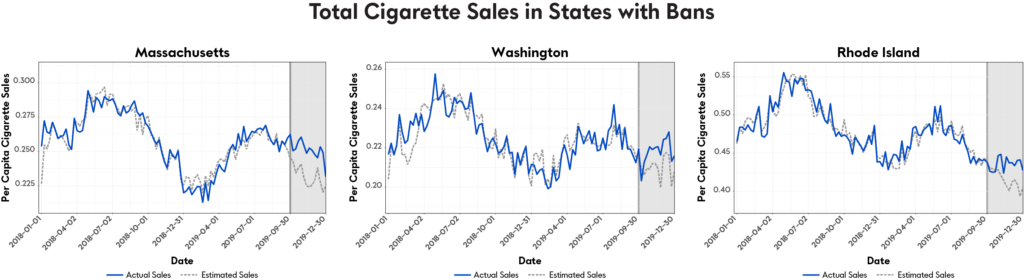
Our population impact team recently shared a new study entitled “The Impact of Banning ENDS Products on Combustible Cigarette Sales: Initial Evidence from U.S. State-Level Policies” at the 3rd Scientific Summit on Tobacco Harm Reduction, which was held virtually. This new research examined the potential consequences of state-level bans on ENDS products, including their potential impact on cigarette sales in those states — in this case, Massachusetts, Washington, and Rhode Island, all of which passed short-term bans on the sale of ENDS in the fall of 2019. Using commercial data on cigarette sales before and after these bans, our researchers modeled the possible impact of these bans on cigarette sales as compared to what would have been observed in the absence of a ban.
They found that cigarette sales in states banning ENDS were significantly higher than would have been observed otherwise. In Massachusetts, for example, where a full ban on ENDS products was passed, weekly per capita cigarette sales were 8.3% higher. In Washington and Rhode Island, where flavored ENDS products were banned, higher than expected cigarette sales occurred — 4.6% in Washington and 5% in Rhode Island.
Overall, researchers estimated that around 3 million additional cigarette packs have been sold during the period of the temporary bans that would not have been sold otherwise, including 1.85 million packs in Massachusetts, 877,000 packs in Washington and 281,000 packs in Rhode Island.
This study provides some of the first evidence that a state’s banning of ENDS products may have unintended consequences, including potentially driving adult smokers back to combustible cigarette use. At the same time, we recognize that the category’s potential will only be realized if we are able to earn a license to operate in society with the trust of regulators, policymakers and other stakeholders. Only if we are a responsible steward of our product and our category and combat underage use, can we help create a world where policies are proportionate to the relative risk of tobacco and nicotine products, thereby ensuring vapor alternatives can compete with combustible cigarettes among adult smokers.
As part of that process, late last year, Juul Labs, under new leadership, committed to resetting the vapor category. We reduced our product portfolio, halted television, print, and digital product advertising, continued to work with states toward full implementation and enforcement of Tobacco 21 and submitted a Premarket Tobacco Product Application to the U.S. Food and Drug Administration for our Tobacco and Menthol products including comprehensive scientific evidence to support the harm reduction potential of our products and data-driven measures to address underage use. We continue to believe that the FDA’s PMTA process is the right forum to determine the role vapor products can play in helping adult smokers move away from combustible cigarettes and support FDA’s active enforcement against illegally marketed products that jeopardize that potential.
Juul Labs will continue to share results from its science and research program transparently with the public health communities as it works to support the scientific basis for the category, as well as future regulatory filings. Future research is needed to evaluate the potential spillover effects of these types of local bans, and to determine the long-term impact of these policies on use of tobacco products and net population health.
About the Study
In this study, researchers utilized Generalized Synthetic Control methods to predict counterfactual cigarette sales for three states that passed temporary bans on ENDS products in 2019: Massachusetts, Rhode Island, and Washington. They compared these counterfactual trends to actual sales in the ban time period in order to assess the impact of these bans on cigarette sales with rigorous causal inference methods.
Overall, the model suggests that the full ban of ENDS in Massachusetts led cigarette sales to be 8.3% (p<0.001) higher than they would have been otherwise. Similar results were seen in states with just flavor bans, ie; in Washington (4.6%, p<0.001) and Rhode Island (5.0%, p<0.001).
Sales of menthol cigarettes were also higher than expected: Actual sales of menthol cigarettes in Massachusetts, Washington and Rhode Island were 12.3%, 6.5% and 7.1% (p<0.001) higher than predicted by their synthetic counterfactuals.
The study, entitled, “The Impact of Banning ENDS Products on Combustible Cigarette Sales: Initial Evidence from U.S. State-Level Policies” was presented at the 3rd Scientific Summit on Tobacco Harm Reduction.
Other Posts
October 25, 2024
ROBYN GOUGELET DELIVERS REMARKS DURING A PUBLIC MEETING HOSTED BY FDA AND NIH
On October 21, Juul Labs’ Vice President of U.S. Regulatory Affairs, Robyn Gougelet, delivered remarks during a joint public meeting hosted by the U.S….
March 17, 2023
JUUL LABS PUBLISHES WHITE PAPER EXAMINING THE REAL-WORLD IMPACT OF ENDS PRODUCTS FOR ADULT SMOKERS
In a new white paper entitled “The Real-World Impact of ENDS for Adult Smokers: Tobacco Harm Reduction Through Real-World Data and Evidence,” Juul Labs has reviewed and compiled the latest science and evidence demonstrating the positive real-world impact of ENDS products for adult smokers.
March 17, 2022
PEER-REVIEWED STUDY FINDS BANNING VAPOR PRODUCTS MAY LEAD TO INCREASED CIGARETTE SALES
This research shows that policies that significantly restrict vapor products are likely deterring current adult smokers from switching and driving former adult smokers back to combustible cigarettes, which remain the leading cause of preventable death in the U.S. and worldwide.